
THE LIVER ENZYME LAB!
*Enzymes are biological catalysts. They help increase the rate of chemical reactions.
Enzymes are most often proteins and their three-dimensional shape is important to their catalytic activity.
Because of their 3-D shape, enzyme are specific for the reactions that they catalyze.
THE REACTION IN THIS EXPERIMENT IS THE DECOMPOSITION OF HYDROGEN PEROXIDE INTO WATER AND OXYGEN GAS! THE REACTION WILL BE CATALYZED BY THE CATALASE ENZYME FOUND IN LIVER.
(because the liver often functions to break down cells)
Purpose:
Hoe do environmental factors affect the activity of an enzyme?
Learn how enzymes function and what affects their rate of reaction and ability to work.
You will measure effects of changes in temperature, PH, and enzyme concentration on reaction rates of an enzyme catalyzed in a controlled experiment.
Hypothesis: As the temperature increases, the initial enzyme reaction will speed up. As the temperature decreases, the initial enzyme reaction will slow down.
(more info in chart)
*Enzymes are biological catalysts. They help increase the rate of chemical reactions.
Enzymes are most often proteins and their three-dimensional shape is important to their catalytic activity.
Because of their 3-D shape, enzyme are specific for the reactions that they catalyze.
THE REACTION IN THIS EXPERIMENT IS THE DECOMPOSITION OF HYDROGEN PEROXIDE INTO WATER AND OXYGEN GAS! THE REACTION WILL BE CATALYZED BY THE CATALASE ENZYME FOUND IN LIVER.
(because the liver often functions to break down cells)
Purpose:
Hoe do environmental factors affect the activity of an enzyme?
Learn how enzymes function and what affects their rate of reaction and ability to work.
You will measure effects of changes in temperature, PH, and enzyme concentration on reaction rates of an enzyme catalyzed in a controlled experiment.
Hypothesis: As the temperature increases, the initial enzyme reaction will speed up. As the temperature decreases, the initial enzyme reaction will slow down.
(more info in chart)
***SAFTEY CONSIDERATION***
Hydrogen peroxide is an oxidizing agent and body tissue irritant. Avoid skin or eye contact. Safety goggles and gloves should be worn when handling hydrogen peroxide. Flush eyes and skin with water immediately and for 15 minutes if you come into contact with hydrogen peroxide. If ingested, do not induce vomiting and call poison control immediately.
Hydrogen peroxide is an oxidizing agent and body tissue irritant. Avoid skin or eye contact. Safety goggles and gloves should be worn when handling hydrogen peroxide. Flush eyes and skin with water immediately and for 15 minutes if you come into contact with hydrogen peroxide. If ingested, do not induce vomiting and call poison control immediately.
Materials
1 molar HCl solution
1 molar NaOH solution
5 test tubes
Measuring Pipette
10 mL graudated cylinder
Scissors and forceps
Stirring Rod
Fresh Liver
Hot Plate
Ice Cooler
Water
1 molar HCl solution
1 molar NaOH solution
5 test tubes
Measuring Pipette
10 mL graudated cylinder
Scissors and forceps
Stirring Rod
Fresh Liver
Hot Plate
Ice Cooler
Water

Procedure
1. Place 2 ml of the 3% hydrogen peroxide
2. Using forceps and scissors cut a small piece of liver and add it to a test tube. The liver should cover the bottom of the tube.
3.Label test tubes #1-5
test tube #1= water
test tube #2= water & liver
test tube #3= water, liver, & add H2O2
test tube #4= water, liver, boiled then H2O2 added
test tube #5= water, liver, cooled then H2O2 added
4. Add each needed material into each test tub as listed above. Cool and heat the livers that must be boiled or chilled, then add H202.
5. Observe reactions.
1. Place 2 ml of the 3% hydrogen peroxide
2. Using forceps and scissors cut a small piece of liver and add it to a test tube. The liver should cover the bottom of the tube.
3.Label test tubes #1-5
test tube #1= water
test tube #2= water & liver
test tube #3= water, liver, & add H2O2
test tube #4= water, liver, boiled then H2O2 added
test tube #5= water, liver, cooled then H2O2 added
4. Add each needed material into each test tub as listed above. Cool and heat the livers that must be boiled or chilled, then add H202.
5. Observe reactions.

*Factors that will effect the effectiveness of enzymes: PH, temperature, and concentration
Independent Variables: temperature of liver & contents in test tube
Dependent variable: height of reaction observed
ENZYME= catalase
Independent Variables: temperature of liver & contents in test tube
Dependent variable: height of reaction observed
ENZYME= catalase

*Indication that the enzyme is working= more bubbles will occur at a faster rate
***CONTROL TEST TUBE= test tube #1****
DATA CHART :
***CONTROL TEST TUBE= test tube #1****
DATA CHART :
|
Test Tube #
|
Contents
|
Hypothesis
|
Observation
|
Interpretations
|
|
1
|
water
|
Nothing will happen, water is just being added in order to have a constant in the experiment.
|
No reaction observed.
|
Constant stayed as a constant with no reaction occurring.
|
|
2
|
water and liver
|
Nothing will happen, water and liver will just remain in the test tube.
|
No reaction observed.
|
No reaction occurred in this test tube.
|
|
3
|
water, liver, add H2O2
|
A reaction will occur due to the H2O2 added.
|
Bubbling occurred when the H2O2 was added to the liver and water, which represented the reaction.
|
Liver has an enzyme called catalase which breaks down hydrogen peroxide (H2O2) into water (H2O) and oxygen (O2). The bubbles you see are oxygen gas.
|
|
4
|
water, liver, boiled, add H2O2
|
A faster reaction will occur due to the higher temperature.
|
A lot of bubbling quicker than observed in test tube 3 without boiling- made reaction occur faster. Test tube is warm = exothermic reaction.
|
The
enzyme activity increases more with the increase of temperature up to a point. |
|
5
|
water, liver, cooled, add H2O2
|
The liver being cooled will slow down the reaction.
|
Slows the reaction.
|
When the liver is cooled, the reaction is slowed due to the lower temperature.
|
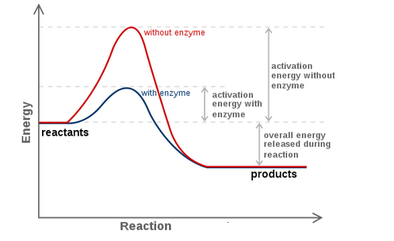
Enzymes speed up initial reaction until it plateaus.
How it relates:
In our cells there are many reactions occurring. One by-product from these reactions is hydrogen peroxide.
*This reaction is important to cells because hydrogen peroxide (H2O2) is produced as a byproduct of many normal cellular reactions.
2 hydrogen peroxide molecules react to form 2 water molecules and 1 oxygen molecule.
Catalase
2H2O2 ⎯⎯⎯⎯→2H2O+O2(gas)
(substrate) (enzyme) (products)
The catalase enzyme reduces the substrate, peroxide, to water and oxygen by the following decomposition reaction.
Conclusion:
All my hypotheses were supported with the data I collected in this lab :)!
This experiment depicts the importance of enzymes in the human body. Enzymes catalyze many important chemical reactions in the body. This demonstration is a hands-on example of how the enzyme catalase, which is present in all cells, but in highest in the liver.
Disposal:
The test tubes that contain only water and liver can be disposed of in regular solid waste, and the liquid can be flushed down the sink with running water.
***Always follow disposal procedures that are consistent with school board protocol and appropriate for your municipality.***
In our cells there are many reactions occurring. One by-product from these reactions is hydrogen peroxide.
*This reaction is important to cells because hydrogen peroxide (H2O2) is produced as a byproduct of many normal cellular reactions.
2 hydrogen peroxide molecules react to form 2 water molecules and 1 oxygen molecule.
Catalase
2H2O2 ⎯⎯⎯⎯→2H2O+O2(gas)
(substrate) (enzyme) (products)
The catalase enzyme reduces the substrate, peroxide, to water and oxygen by the following decomposition reaction.
Conclusion:
All my hypotheses were supported with the data I collected in this lab :)!
This experiment depicts the importance of enzymes in the human body. Enzymes catalyze many important chemical reactions in the body. This demonstration is a hands-on example of how the enzyme catalase, which is present in all cells, but in highest in the liver.
Disposal:
The test tubes that contain only water and liver can be disposed of in regular solid waste, and the liquid can be flushed down the sink with running water.
***Always follow disposal procedures that are consistent with school board protocol and appropriate for your municipality.***

